The past decade brought tremendous gains in life expectancy. Globally, life expectancy at birth increased from 47 years in 1950 to 73 years in 2020 and is projected to continue to grow to 77 years by 2050 (UN Department of Economic and Social Affairs 2019). With this growth in life expectancy has come the recognition that public health efforts need to focus on improving the quality of life in these additional years.
Thus, the UN declared 2021–2030 the ‘Decade of Healthy Ageing’. With this declaration, the UN and the World Health Organization set measurable targets to optimise five areas of functioning among older adults over the next ten years: “the ability to meet one’s basic needs; the ability to learn, grow and make decisions; mobility; the ability to build and maintain relationships; and the ability to contribute” (World Health Organization 2021).
Alzheimer’s disease and related dementias (ADRDs) pose one of the greatest threats to the goals of the Decade of Healthy Ageing.1 ADRDs also offer a critical opportunity to optimise functioning, if the global community invests now. While the onset of ADRDs typically occurs in midlife, symptoms generally become more severe as people age.2 As a result, cognitive and other domains of functioning decline, causing suffering for individuals, their families, and their communities.
Despite these cascading effects, to date, no effective therapies have been developed for ADRDs (although recent clinical trials have produced some potentially exciting early results; see Eli Lilly and Company 2021). Here, we demonstrate the immense opportunity to gain not only years lived but years lived well, by investing in ADRDs treatment.
We forecast the global disease burden of ADRDs, using data from the Institute for Health Metrics and Evaluation for 2010–2019 (Carroll 2019). We compute per-person growth rates of disability-adjusted life years (DALYs) – a standard measure of the burden of disease3 – due to ADRDs, by country and age group.
Note that using DALYs in resource-allocation decisions has been criticised for discriminating against certain populations – including a critique in the context of the UN Decade of Healthy Ageing for DALYs’ discrimination against older adults (Bloom et al. 2021). Diseases in early life frequently produce more DALYs than those in later life because of differences in remaining life expectancy, so resource decisions based on DALYs are heavily weighted against investment in older adults.
The Decade of Healthy Ageing reflects a normative commitment that the rights and dignity of older adults require equitable attention and investment, beyond what DALYs alone indicate. To reflect this issue of equity across the life course, we specifically focus our DALY comparisons on the population over the age of 55 and encourage decisionmakers to consider other key normative commitments when allocating scarce health resources, aside from the allocation suggested by DALYs alone.
In our calculations, we assume that the per-person disease burden will grow at the same rate in future years, and we project country-level DALYs through 2050 based on population counts and age structure data from the UN’s World Population Prospects. The estimates are adjusted upward to include dementias related to Alzheimer’s disease (e.g. vascular dementia) that are not available from the Institute for Health Metrics and Evaluation (the Institute’s ‘Alzheimer’s disease and other dementias’ classification does not include all ADRDs conditions).
We calculated the adjustment factor from age-specific prevalence rates of ADRDs in the US Aging, Demographics and Memory Study, which is based on a sample of respondents from the US Health and Retirement Study (Hudomiet et al. 2018). We estimate DALYs from ADRDs in five-year age groups for the population over the age of 40 in each country. Those over the age of 80 in a country are combined into one group.
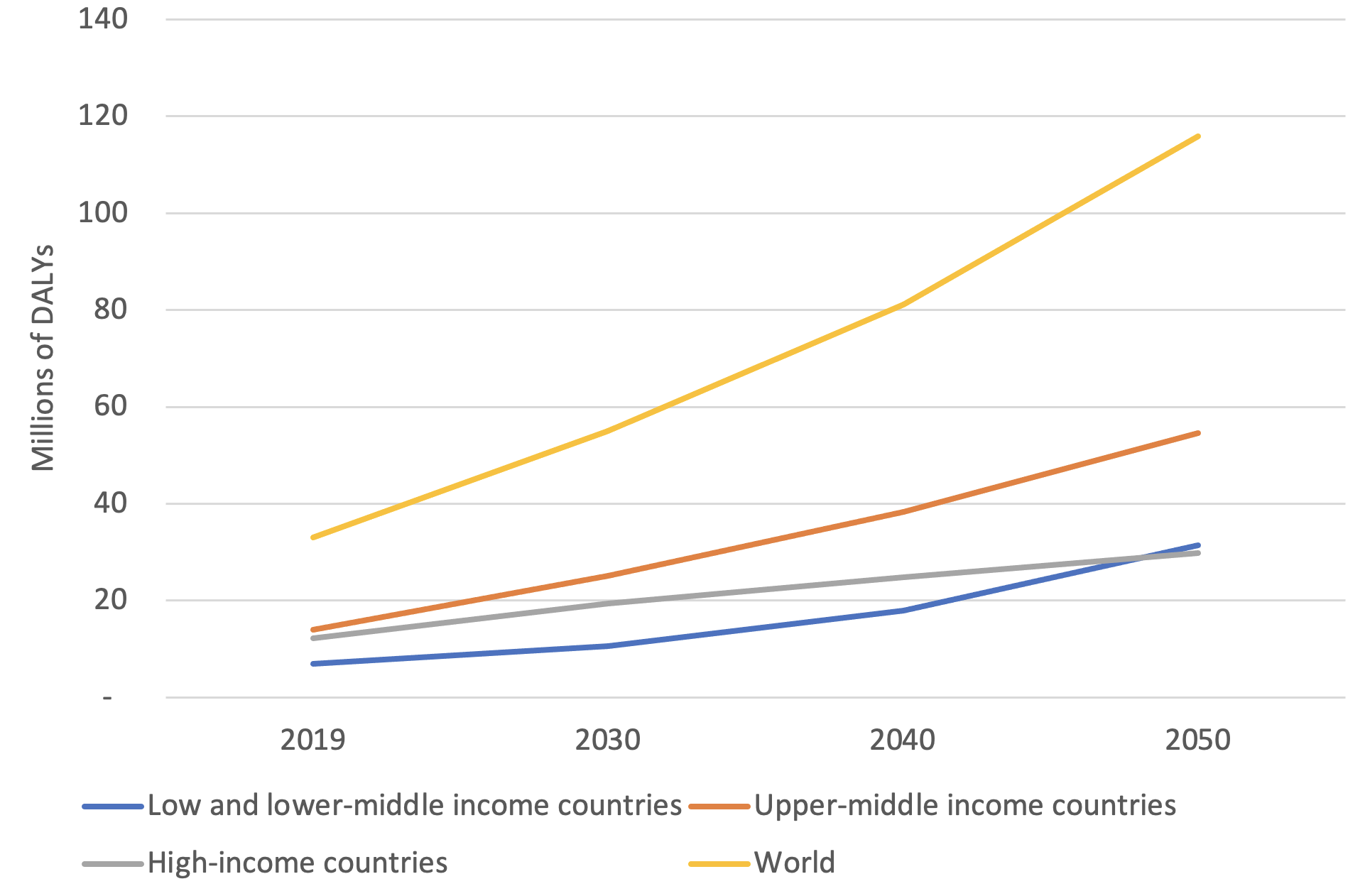
Figure 1 presents our estimates aggregated by the World Bank classification of country income level in 2020. We find that ADRDs contributed 33.1 million DALYs in 2019, and if the burden continues to grow in line with our model, it will contribute 55.1 million DALYs in 2030, 81.1 million in 2040, and 115.8 million in 2050, indicating that the global burden of ADRDs will more than triple over the next 30 years.
Figure 1 Estimated DALYs attributable to ADRDs, 2019–2050
Notes: Analysis includes 192 countries for which data are available, covering more than 99% of the world’s population. Country income group classifications are based on the World Bank’s 2020 definition.
Further, the distribution of the disease burden of ADRDs will shift over time: the share of ADRD DALYs in low- and lower-middle-income countries is projected to grow by 29% between 2019 and 2050, while the share in upper-middle-income countries is projected to grow only 12% and the share in high-income countries will decrease by 30% (a product of lessening, but still vast, disparities in life expectancy among countries). By 2050, low- and lower-middle-income countries are projected to contribute more DALYs associated with ADRDs than their high-income counterparts.
Compared with other health conditions measured by the Institute for Health Metrics and Evaluation, ADRDs were the sixth greatest contributor to DALYs globally for people aged 55 and over in 2019. Using the same analytic approach to forecast the DALY burden of the other highest-burden diseases, we estimate that ADRDs will become the fifth greatest DALY contributor for people aged 55 and over by 2050, overtaking chronic obstructive pulmonary disorder.
A rapidly growing economic burden will also accompany the growing ADRD disease burden, as ADRDs impair individual functioning and demands intensive caregiving and related support. For example, one recent study forecasts the global economic costs associated with dementia (which includes ADRDs and other causes of dementia), including medical care, long-term care and related services, and foregone wages for both the individual and caregivers (Jia et al. 2018). It estimates that total dementia spending reached $1.33 trillion annually in 2020 and will climb to $2.54 trillion in 2030, $4.83 trillion in 2040, and $9.12 trillion in 2050, meaning that the costs will double almost every decade that the ADRD burden goes unmitigated.
Moreover, these forecasts and others from the literature actually underestimate the full economic burden of ADRDs (Wimo et al. 2017, El-Hayek et al. 2019). Although they capture much of the direct spending in easily monetisable areas, they often struggle to fully account for spillovers onto the larger economy, the magnitude of opportunity costs for informal caregivers, productive nonmarket activities, or the total value of suffering across those impacted. Unless mitigated through effective investments in research, diagnosis, treatment, support, and prevention, ADRDs will create a substantial drag on global economic growth and exacerbate global economic inequities in the coming decades.
Failing to account for the full economic and societal burden of ADRDs leads to underinvestment in supportive interventions that ease the impact of these diseases and in R&D for therapeutics that could address ADRDs. While we lack effective therapies for ADRDs, there is strong evidence for supportive interventions that improve quality of life and reduce the toll of these diseases for those affected (Walter and Pinquart 2020, Kim and Park 2017). Scaling those interventions and improving on them remains an imperative and an important equity concern, as those with the fewest resources have the least access to them.
Further, COVID-19 has demonstrated that, as a global community, we can tackle the most complex research and discovery challenges rapidly and effectively when the economic peril of inaction is obvious, and we invest sufficiently to address the issue. A combination of private investment, public funding, cross-national collaboration, and multilateral commitments allowed the global research community to accelerate discovery by, among other strategies, creatively applying experimental biological interventions (such as RNA therapies, which had shown promise for treating cancer) to curb the new pandemic (Sampat and Shadlen 2021).
A pandemic like COVID-19 had been considered a low-probability event of unknown human and economic impact. Within one year, it became a reality, and given the potential costs (in millions of lives and trillions of dollars), funding was unlocked and billions of doses of multiple vaccines are being rolled out.
Unlike the current pandemic, the overwhelming burden of ADRDs is a clear high-probability event. The global community can learn from the collective investment and coordinated effort that led to breakthroughs in the COVID-19 pandemic to similarly increase the likelihood of treatment advances for ADRDs and avert a portion of its associated massive health and economic tolls.
Admittedly, COVID-19 and ADRDs are an imperfect analogy – ADRDs are a more challenging area of research that make breakthroughs less likely. ADRDs, like many central nervous system disorders, face distinct R&D difficulties, including an insufficient understanding of underlying disease mechanisms, challenges with clinical trial recruitment, and difficulty with preclinical models (Goldman et al. 2018, Gauthier et al. 2016).
On the other hand, cancer certainly presents a strong comparison as an example of an extremely difficult area of R&D with a very low likelihood of success. Because of substantial investment, cancer R&D yielded critical breakthroughs that alleviate suffering and add years of life (Wong et al. 2019). Analysis of drug, biological, or device-oriented interventional clinical trials listed in ClinicalTrials.gov on 15 December 2020 found approximately 50 times as many trials were underway for cancer as for ADRDs – a notable disparity as cancer only accounts for approximately eight times as many DALYs as ADRDs. If ADRDs received investment proportionally comparable to cancer, we would likely see accumulating treatment breakthroughs even with a low probability of success.
To ensure the future health and functioning of the ageing global community, we must learn from the lessons of the COVID-19 pandemic and invest effectively and efficiently in ADRD R&D, along with scaling effective supportive interventions, to mitigate the impending health, economic, and social burdens of ADRDs.
These investments could also reduce global health and economic inequities, as the burden of these diseases will increasingly fall on low- and middle-income countries. With coordinated global action to advance ADRD care and treatment, we will be able to address a key contributor to functional decline in older adults and fulfil the promise of the UN’s Decade of Healthy Ageing.
References
Bloom, D E, D Cadarette, M Ferranna, R N Hyer and D L Tortorice (2021), “How new models of vaccine development for COVID-19 have helped address an epic public health crisis”, Health Affairs 40(3): 1–8.
Carroll, W M (2019), “The global burden of neurological disorders”, The Lancet Neurology 18(5): 418–9.
El-Hayek, Y H, R E Wiley, C P Khoury, R P Daya, C Ballard, A R Evans, M Karran, J L Molinuevo, M Norton and A Atri (2019), “Tip of the iceberg: Assessing the global socioeconomic costs of Alzheimer’s disease and related dementias and strategic implications for stakeholders”, Journal of Alzheimer’s Disease 70(2): 323–41.
Eli Lilly and Company (2021), “Lilly’s Donanemab slows clinical decline of Alzheimer’s disease in positive Phase 2 trial”, Cision PR Newswire, 11 January.
Gauthier, S, M Albert, N Fox, M Goedert, M Kivipelto, J Mestre-Ferrandiz and L: Middleton (2016), “Why has therapy development for dementia failed in the last two decades?”, Alzheimer’s and Dementia 12(1): 60–4.
Goldman, D P, H Fillit and P Neumann (2018), “Accelerating Alzheimer’s disease drug innovations from the research pipeline to patients”, Alzheimer’s and Dementia 14(6): 833–6.
Hudomiet, P, M D Hurd and S Rohwedder (2018), “Dementia prevalence in the United States in 2000 and 2012: Estimates based on a nationally representative study”, The Journals of Gerontology Series B: Psychological Sciences and Social Sciences 73(suppl.1): S10–S19.
Jia, J, C Wei, S Chen, F Li, Y Tang, W Qin, L Zhao, H Jin, H Xu, F Wang, A Zhou, et al. (2018), “The cost of Alzheimer’s disease in China and re‐estimation of costs worldwide”, Alzheimer’s and Dementia 14(4): 483–91.
Kim, S K, and M Park (2017), “Effectiveness of person-centered care on people with dementia: A systematic review and meta-analysis”, Clinical Interventions in Aging 12: 381.
Nicholas, L H, K M Langa, J P Bynum and J W Hsu (2021), “Financial presentation of Alzheimer disease and related dementias”, JAMA Internal Medicine 181(2): 220–7.
Sampat, B N, and K C Shadlen (2021), “The COVID-19 innovation system”, Health Affairs 40(3).
United Nations Department of Economic and Social Affairs (2021), World population prospects 2019.
Walter, E, and M Pinquart (2020), “How effective are dementia caregiver interventions? An updated comprehensive meta-analysis”, The Gerontologist 60(8): e609–19.
Wimo, A, M Guerchet, G-C Ali, Y-T Wu, A M Prina, B Winblad, L Jönsson, Z Liu and M Prince (2017), “The worldwide costs of dementia 2015 and comparisons with 2010”, Alzheimer’s and Dementia 13(1): 1–7.
Wong, C H, K W Siah and A W Lo (2019), “Estimation of clinical trial success rates and related parameters”, Biostatistics 20(2): 273–86.
World Health Organization (2021), Decade of Healthy Ageing: Baseline report.
Endnotes
1 Within ‘Alzheimer’s disease and related dementias’, we follow the US National Institutes of Health classification of Alzheimer’s disease and related dementias, which includes Alzheimer’s disease, frontotemporal dementia, Lewy body dementia, vascular contributions to cognitive impairment and dementia, and mixed etiology dementias, which all involve progressive degeneration of cognitive functioning (https://www.ninds.nih.gov/Current-Research/Focus-Disorders/Alzheimers-Related-Dementias).
2 Evidence increasingly indicates that Alzheimer’s disease and related dementias have effects years before diagnosis (e.g. Nicholas et al. 2021).
3 One DALY represents the loss of the equivalent of one year of full health (World Health Organization, https://www.who.int/data/gho/indicator-metadata-registry/imr-details/158).