In 2012, the International Agency for Research on Cancer (IARC) estimated that there were 14.1 million new cancer cases and 8.2 million cancer deaths worldwide. Over the past decade, exciting breakthroughs in thefield of immuno-oncology have resulted in significant gains in long-term survival.1 A major drawback is that the cost of immunotherapy is often extremely high. The combination of Yervoy (ipilimumab) and Opdivio (nivolumab), used in the treatment of melanoma, costs $159,100 for a standard 12-week course. The cost of the CAR-T cell therapy Kymriah is $475,000. In this column, we leave the important but thorny issue of drug pricing aside and instead focus on financing solutions for life-extending treatments such as immunotherapy.
Existing solutions to finance expensive life-extending treatments have important shortcomings. In the US, health insurance plans typically come with copays to limit moral hazard. Patients may not be able to afford the copays in addition to the cost of insurance. When healthcare is tied to employment, job loss exposes patients to reclassification risk. For those without employer-provided health insurance, the out-of-pocket cost for treatment under the Affordable Care Act (ACA) often exceeds 30% of pre-tax income.2 Gupta et al. (2015) find that cancer diagnoses increase default and foreclosure rates, in part due to incomplete insurance coverage.
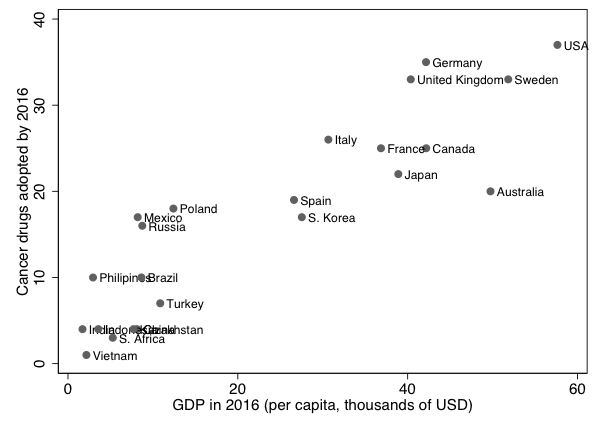
Outside of the US, governments tend to play a more important role in health insurance. Facing ever tighter budgets and mounting debt, governments can ill afford the cost of the expensive life-extending treatments (minus the copay). If we look at the adoption of new cancer treatments across countries, we find a strong correlation between the adoption rate and a country’s per-capita income, see Figure 1. For instance, only 20 of the 42 immunotherapy drugs developed between 2011 and 2015 were available in Spain and South Korea in 2016. While other factors beyond fiscal constraints may also be at play, the figure highlights the need to accelerate the widespread adoption of new immunotherapies.
In response, credit-based solutions have been proposed (Montazerhodjat et al. 2016). But households cannot pledge their future labour income and may default on loans received for medical treatment. Higher earnings uncertainty after treatment further limits the borrowing capacity.
Figure 1 Number of cancer drugs launched and GDP
Note: The figure plots the number of cancer drugs launched between 2011 and 2015 that are available across countries (vertical axis) relative to the 2016 GDP per capita in USD.
Data source: QuintilesIMS Institute (2017).
Under the status quo, we face a future where life-saving treatments are effectively unavailable for a large segment of the population. The conundrum will only get worse as (i) the world population ages and with it the incidence of cancer; (ii) immunotherapies become more effective, approved for more cancer sites and increasingly as a first-line therapy, and are applied at earlier stages of the disease; and (iii) the fiscal position of governments all over the world worsens.
The benefit to life insurers of immunotherapy
There exists a powerful solution to this funding crisis. Our main insight is that life insurance companies experience large benefits from immunotherapies. An example illustrates the basic idea.
Consider an individual who purchases a life policy at age 30 and is diagnosed with stage-4 melanoma at age 40. Per dollar of death benefit (or face value), the policy now has a present value of -$0.95 to the insurer. Clinical studies of stage-4 melanoma indicate a 50% probability of durable survival with immunotherapy. The expected gain in survival raises the value of the life insurance contract to the insurer to -$0.51. The insurer's benefit from immunotherapy is therefore $0.44 per dollar of face value. A policy with a death benefit of $362,000 would generate a benefit that is the same as the entire $159,000 cost of the state-of-the-art Yervoy plus Opdivio treatment. A patient would typically face ‘only’ the copay and maximum out-of-pocket costs, about $20,000 for the typical family on Obamacare. A life insurance policy with a face value as small as $46,000 generates a benefit large enough to cover these out-of-pocket costs.
Importantly, many households own life insurance policies. Among all financial instruments (stocks, bonds, annuities, etc.) it enjoys the highest participation rate, with 68% of men between age 35 and 54 owning life insurance and 63% of women in 2016. The average death benefit for individuals between 35 and 44 years of age is $240,937, far exceeding the minimum necessary benefit of $46,000 in the above example.
Several parameters determine the benefit of a new life-extending treatment to life insurers: the increase in survival probability resulting from treatment, the typical age of diagnosis, and patient demographics (in particular age and sex). In a recent paper (Koijen and Van Nieuwerburgh 2018), we provide a life insurance valuation model with these ingredients. We compile evidence on these parameters and quantify the insurers' benefit from immunotherapy. We provide detailed estimation for the case of stage-4 melanoma using data on incidence and mortality by age and gender, as well as evidence from clinical trials on immunotherapy.
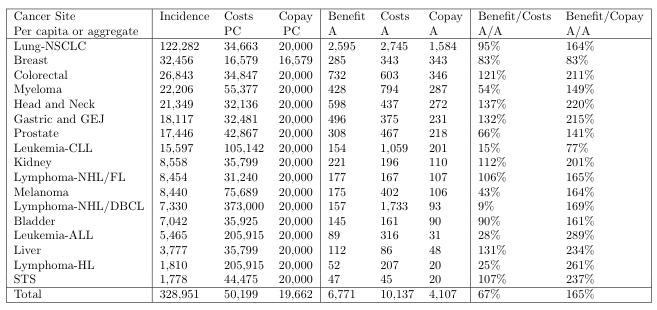
Table 1 extends this evidence to all 17 cancer sites with FDA-approved immunotherapies. Our calculations show that the aggregate benefit from immunotherapies to life insurers is about $6.8 billion per year. For comparison, the total cost of providing immunotherapy treatment to those patients that own life insurance is $10.1 billion per year. Their aggregate copay is $4.1 billion. In other words, the aggregate benefit amounts to two-thirds of the aggregate cost of treatment and is more than enough to provide for the patients’ share of the cost (167%). This underscores the potential funding that can be unlocked.
Table 1
Solutions to finance the war on cancer
The large benefits to life insurers raise questions about the boundaries between health and life insurance markets going forward. An integrated insurer would offer higher coverage rates for life-extending treatments, internalising the benefit to the life insurance arm.
However, our focus is on solutions that can be implemented immediately without further delay.
We discuss different financing mechanisms that vary depending on the bargaining power between the insurance company and consumers. If consumers have all bargaining power, then the insurer covers the $4.1 billion copay and still earns $2.7 billion from the immunotherapy breakthroughs. This constitutes a Pareto improvement, a rare example of a free lunch. First, the patient gains access to treatment and the benefits of a longer life, while maintaining financial stability. She would not have to spend most of their savings on treatment nor have to lapse or sell her life insurance policy. She can resume life after cancer in good financial as well as physical health. If and when she dies, the life insurance policy would serve its intended purpose of providing financial stability for her dependents.
If the insurer has all the bargaining power instead, it can allow consumers to borrow against their death benefit at fair value to finance immunotherapies. Or it can allow consumers to use the death benefit for treatment, reducing the death benefit by the cost of treatment. The former solution uses the increased death benefit tied up in the insurance company as collateral, thereby bringing credit solutions within reach for a large group of patients. The latter solution corresponds to a perfectly efficient life settlement market, a secondary market place for life insurance policies. There exists a life settlement market on which investors buy policies from sick policy holders, but often at deep discounts (Fang and Kung 2017). Traditional life settlements suffer from the additional drawback that the buyer of the policy has a financial incentive for the patient to die as soon as possible, a misalignment of incentives. In our solution, the incentives of the life insurer and the patient remain perfectly aligned.
Regardless of the precise bargaining power, there would be enormous gains in reputation for life insurance companies from saving lives. Furthermore, life insurance would become a more valuable product to consumers because it would now pay for life-enhancing medical treatment in case of a cancer diagnosis. Widespread adoption of this funding model would increase life expectancy in the population, which would lower the cost of life insurance. The life insurance market would grow for all these reasons. A virtuous cycle of more life insurance premium revenue, higher life insurance participation rates and coverage, and more payments for treatment would result. A larger drug market would stimulate further development of immunotherapies, accelerating the virtuous cycle.
References
Fang, H and E Kung (2017), “Life Insurance and Life Settlements: The Case for Health-Contingent Cash Surrender Values”, Working Paper, University of Pennsylvania.
Gupta, A, E R Morrison, C R Fedorenko, and S Ramsey (2015), “Cancer Diagnoses and Household Debt Overhang,” Working Paper, NYU Stern.
Koijen, R and S Van Nieuwerburgh (2018), “Financing the War on Cancer,” Working Paper, NYU Stern.
Montazerhodjat, V, D M Weinstock, and A W Lo (2016), “Buying Cures Versus Renting Health: Financing Health Care with Consumer Loans,” Science Translational Medicine 8(327): 1–7.
Endnotes
[1] Immunotherapy refers to a set of treatments that stimulate the body's immune system to attack cancer cells. The American Cancer Society distinguishes between five categories of immunotherapies: (i) monoclonal antibodies, (ii) immune checkpoint inhibitors, (iii) adoptive cell therapies, (iv) cancer vaccines, and (v) cytokines. Over the past five years, the largest number of new drugs were immune checkpoint inhibitors. PD-1/PD-L1 and CTLA-4 are examples of checkpoint proteins that sit on the surface of the cancer cells and tell the T cells to leave the cancer alone. Immune checkpoint modulators interrupt this signal and unmask the cancer so T cells recognise it and activate. Unlike traditional cytotoxic chemotherapies and radiation, immunotherapies are fairly well tolerated, leave the healthy cells unscathed, can be repeated indefinitely, resulting in a more durable response.
[2] For example, the Silver plan under the ACA, commonly known as ObamaCare, provides health care that costs $5,500 per year in premiums in 2018 for a family of four with two children and earning the average US household income of $65,000 a year. The maximum out-of-pocket costs are $14,700 for this plan. The combined $20,200 amounts to 31% of pre-tax income. For a household earning $100,000, the insurance premiums are $18,300, and the total cost at the maximum out-of-pocket level are $33,000 or 33% of pre-tax income.