Increased longevity is an important part of economic growth, broadly defined. Nordhaus (2005) argued that “to a first approximation, the economic value of increases in longevity in the last hundred years is about as large as the value of measured growth in non-health goods and services”. There is also consensus among macroeconomists that technological progress is the principal source of GDP growth. Romer (1990) argued that “growth…is driven by technological change that arises from intentional investment decisions made by profit-maximizing agents”, while Jones (2002) argued that “long-run growth is driven by the discovery of new ideas throughout the world.” Since technological progress, or the discovery of new ideas, is the fundamental source of one of the major components – GDP growth – of economic growth, broadly defined, it is quite plausible that the discovery of new ideas has played a major role in increased longevity.
The impact of new ideas on mortality
In a recent paper (Lichtenberg 2018a), I test the hypothesis that the arrival of new medical ideas has played a major role in the long-run increase in cancer survival and the decline in cancer mortality in the US, by investigating whether the types of cancer that have been subject to greater penetration of new ideas experienced larger gains in survival and reductions in mortality, controlling for changing incidence. I allow for substantial lags in the relationship between new ideas and cancer patient outcomes, as numerous case studies have indicated that it takes a long time for evidence from research to reach clinical practice. Cancer is the leading cause of an important measure of mortality, namely, the number of years of potential life lost before the age of 80. Thanks to the existence of cancer registries, I am able to control for changes in the number of people diagnosed and their characteristics, such as their mean age.
To construct measures of the penetration of new medical ideas applied to most types of cancer over time, I use the US National Library of Medicine’s MEDLINE/PubMED database, which contains more than 23 million references to journal articles published since 1946 in 5,400 of the world's leading biomedical journals. MEDLINE/PubMED records are indexed with Medical Subject Headings (MeSH), which the National Library of Medicine claims is “one of the most highly sophisticated thesauri in existence today”.1 Between 1955 and 2015, the number of MeSH descriptors increased from 15,800 to 27,800. New descriptors are assigned to “research articles” rather than to “non-research” articles.
My estimates indicate that the five-year survival rate is not related to the novelty of ideas of articles (as measured by their fraction of post-1975 MeSH descriptors) published 0-9 years earlier, but it is strongly positively related to the novelty of ideas in articles published 12-24 years earlier. This finding is consistent with evidence on the lag in research evidence reaching clinical practice. Between 1994 and 2008, the five-year observed survival rate for all types of cancer combined increased from 52.1% to 61.2%. The estimates suggest that about 70% of this increase may have been due to the increase in the novelty of medical ideas 12-24 years earlier.
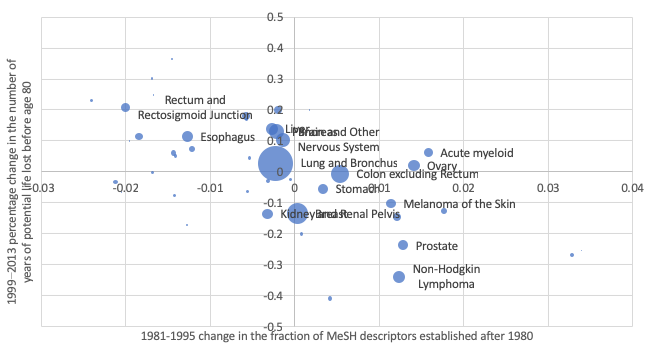
The number of years of potential life lost from cancer before the ages 80 and 70 and the number of cancer deaths are inversely related to the novelty of ideas in articles published 12-24 years earlier, conditional on the number of patients diagnosed 1-10 years before and their mean age at the time of diagnosis. Figure 1 shows the relationship across types of cancer between the 1981–1995 change in the fraction of MeSH descriptors established after 1980 and the 1999–2013 percentage change in the number of years of potential life lost from cancer before age 80, controlling for changes in incidence. The increase in descriptor novelty is estimated to have caused a 38% decline in the premature (i.e. before the age of 80) cancer mortality rate 12-24 years later. The fact that this is much larger than the actual reduction in the premature cancer mortality rate (at 8%) may be due, in part, to declining competing risk from cardiovascular disease.
Figure 1 Relationship across cancer sites between the 1981-1995 change in the novelty of medical ideas and the 1999-2013 percentage change in premature (before age 80) cancer mortality, controlling for changes in incidence
Note: Bubble size is proportional to mean premature (before age 80) cancer mortality.
The impact of new cancer drugs on cancer mortality
In the study described above I analyse the impact of biomedical innovation in general on cancer survival and mortality in a single country (the US). In another recent paper (Lichtenberg 2018b), I focus on the impact of a specific, but very important, type of biomedical innovation – new cancer drugs – on cancer mortality in 36 countries. In 2007, 89% of private biomedical research expenditure was funded by pharmaceutical and biotechnology firms, with the remaining 11% funded by medical device firms (Dorsey et al. 2010).
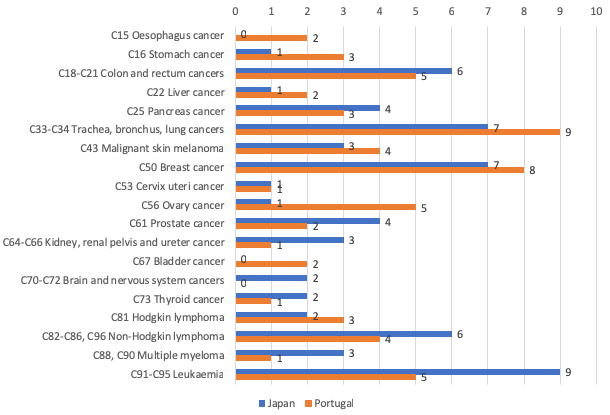
I analyse the correlation across countries between relative mortality from 19 types of cancer in 2015 and the relative number of drugs previously launched in that country to treat that type of cancer, controlling for relative incidence. This approach is feasible because the relative number of drugs launched for different types of cancer has varied considerably across countries. This is illustrated in Figure 2, which shows the number of drugs launched from 2006 to 2015 in Japan and Portugal for 19 types of cancer. Across all types of cancer, the mean number of drugs launched during 2006-2015 was almost identical in Japan and Portugal (3.3 and 3.2, respectively), but Japan launched four more drugs for leukaemia and four fewer drugs for ovary cancer. In the paper I show that the relative number of drugs launched for a particular type of cancer in a country is positively related to relative market size (i.e. the number of patients diagnosed).
Figure 2 Number of drugs launched during 2006-2015 in Japan and Portugal for 19 types of cancer
The number of disability-adjusted life years (DALYs) lost is unrelated to drug launches 0-4 years earlier. This is not surprising, since utilisation of a drug tends to be quite low during the first few post-launch years. Moreover, there is likely to be a lag of several years between utilisation of a drug and its impact on mortality. However, mortality is significantly inversely related to the number of drug launches at least five years earlier, especially to drug launches between five and nine years earlier. One drug launched during 2006-2010 is estimated to have reduced the number of DALYs in 2015 due to the relevant type of cancer by 5.8%; another drug launched during 1982-2005 is estimated to have reduced the number of DALYs in 2015 by about 2.6%. Lower quality (or effectiveness) of earlier-vintage drugs may account for their smaller estimated effect.
The estimates imply that drugs launched during 2006-2010 reduced the number of cancer DALYs in 2015 by about 8.7%, and that had there been no new drug launches during 2006-2010, there would have been 8.04 million additional DALYs due to cancer in the 36 countries covered in my study. The estimates also imply that, in the absence of new drug launches during 2006-2010, there would have been an additional 4.51 million years of life lost before the age of 75, and 2.52 million additional years of life lost before the age of 65.
I also estimate that drugs launched during the entire period of 1982 to 2010 reduced the number of cancer DALYs in 2015 by about 23.0%, and that had there been no new drug launches during 1982-2010, there would have been additional 26.3 million DALYs in 2015. Furthermore, the nine countries with the largest number of drug launches during 1982-2010 are estimated to have had 14% fewer cancer DALYs (controlling for incidence) in 2015 than the nine countries with the smallest number of drug launches during 1982-2010.
Estimates of the cost per life-year gained in 2015 from drugs launched during 2006-2010 ranged between $1,635 (for life-years gained at all ages) and $2,820 (for life-years gained before age 65). These estimates are similar to those obtained in previous country-specific studies in Belgium, Canada, and Mexico, and are well below the estimate obtained in one study of Switzerland.
Mortality in 2015 is strongly inversely related to the number of drug launches in 2006-2010. If a similar relationship holds between mortality in 2020 and the number of drug launches in 2011-2015, the reduction in mortality between 2015 and 2020 due to drug launches that took place 5-9 years earlier (9.9%) will be even greater than the 8.4% reduction between 2010 and 2015.
References
Dorsey, E R (2010), “Financial Anatomy of Biomedical Research, 2003 – 2008”, JAMA 303(2): 137–143.
Jones, C I (2002), “Sources of U.S. Economic Growth in a World of Ideas”, American Economic Review 92(1): 220-239.
Lichtenberg, F R (2018a), “The long-run impact of new medical ideas on cancer survival and mortality”, NBER Working Paper No. 25328 (also published online in Economics of Innovation and New Technology).
Lichtenberg, F R (2018b), “The Impact of New Drug Launches on Life-Years Lost in 2015 from 19 Types of Cancer in 36 Countries”, NBER Working Paper No. 24536 and Journal of Demographic Economics 84(03): 309-54.
Nordhaus, W D (2005), “Irving Fisher and The Contribution of Improved Longevity to Living Standards”, American Journal of Economics and Sociology 64(1): 367-392.
Romer, P M (1990), “Endogenous Technological Change”, Journal of Political Economy 98(5, Pt. 2): S71–S102.
Endnotes
[1] Source: https://www.nlm.nih.gov/mesh/meshrels.html